2025
Reply to Kaestner et al.: Activation of PIEZO1 is not significant for the passage of red blood cells through biomimetic splenic slits
Alexis Moreau, François Yaya, Huijie Lu, Anagha Surendranath, Anne Charrier, Benoit Dehapiot, Emmanuèle Helfer, Annie Viallat, Zhangli Peng
Proceedings of the National Academy of Sciences of the United States of America 122 (2025)10.1073/pnas.2411469121
Dual role of Xenopus Odf2 in multiciliated cell patterning and differentiation
Aude Nommick, Alexandre Chuyen, Raphaël Clément, Virginie Thomé, Fabrice Daian, Olivier Rosnet, Fabrice Richard, Nicolas Brouilly, Etienne Loiseau, Camille Boutin, Laurent Kodjabachian
Developmental Biology 520:224-238 (2025)10.1016/j.ydbio.2025.01.014
Fluid Mechanics of Blood Cells and Vesicles Squeezing Through Narrow Constrictions
Zhangli Peng, Annie Viallat, Yuan-Nan Young
Annual Review of Fluid Mechanics 58:331-354 (2025)10.1146/annurev-fluid-112723-052727
A novel red blood cell deformability biomarker is associated with hemolysis and vaso-occlusive crises in sickle cell disease
Maxime Sahun, Emmanuelle Bernit, Scott Atwell, Alexander Hornung, Anne M Charrier, Imane Agouti, Nathalie Bonello-Palot, Mathieu Cerino, Emmanuèle Helfer, Catherine Badens, Annie Viallat
Scientific Reports 15:15864 (2025)10.1038/s41598-025-00152-w
2024
Dans les bronches, cils et mucus collaborent pour protéger nos voies aériennes
Étienne Loiseau, Annie Viallat
Reflets de la Physique 14-17 (2024)10.1051/refdp/202479014
2023
Transit Time Theory for a Droplet Passing through a Slit in Pressure-Driven Low Reynolds Number Flows
Spencer W Borbas, Kevin Shen, Catherine Ji, Annie Viallat, Emmanuèle Helfer, Zhangli Peng
Micromachines 14:2040 (2023)10.3390/mi14112040
Classification of red cell dynamics with convolutional and recurrent neural networks: a sickle cell disease case study
Maxime Darrin, Ashwin Samudre, Maxime Sahun, Scott Atwell, Catherine Badens, Anne Charrier, Emmanuèle Helfer, Annie Viallat, Vincent Cohen-Addad, Sophie Giffard-Roisin
Scientific Reports 13:745 (2023)10.1038/s41598-023-27718-w
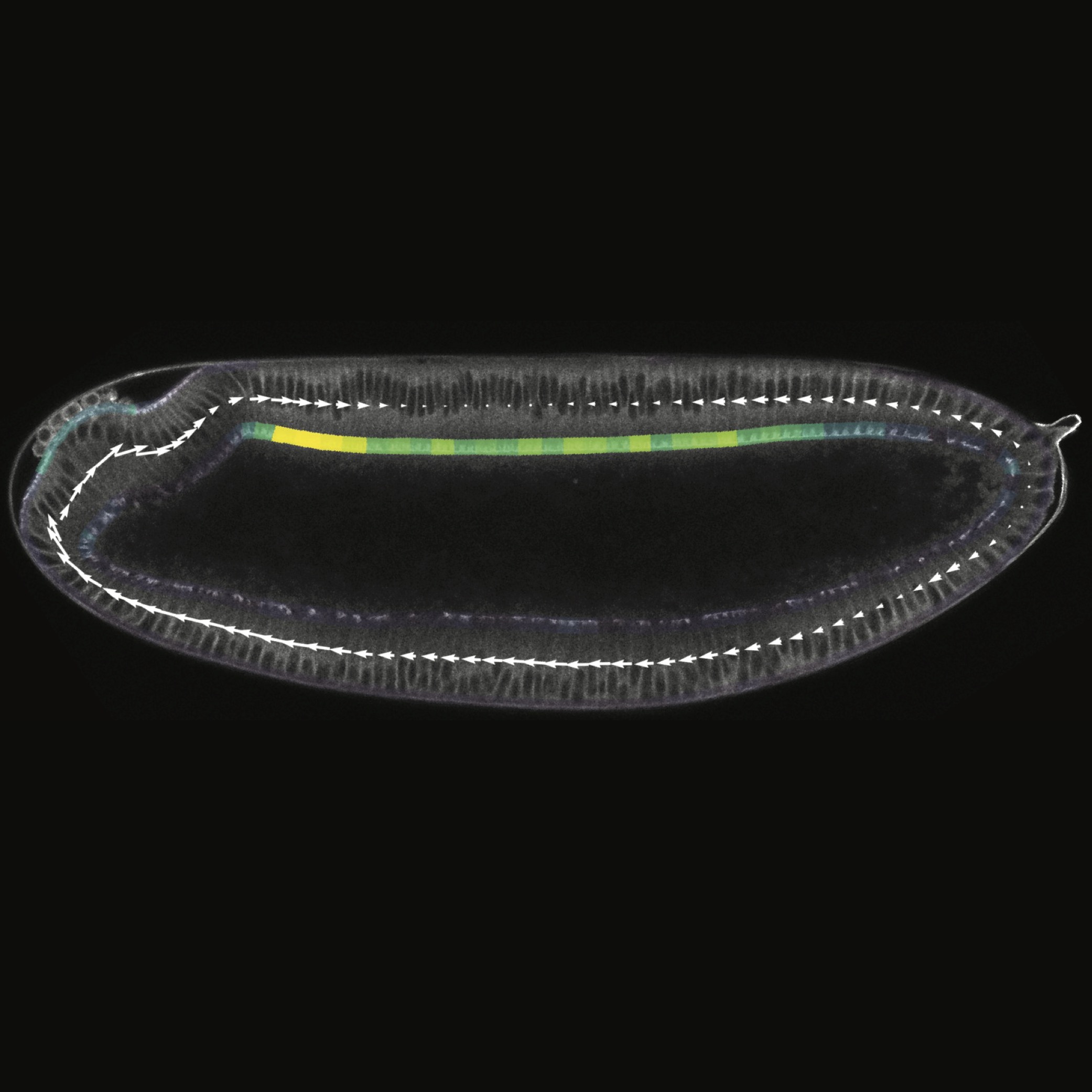
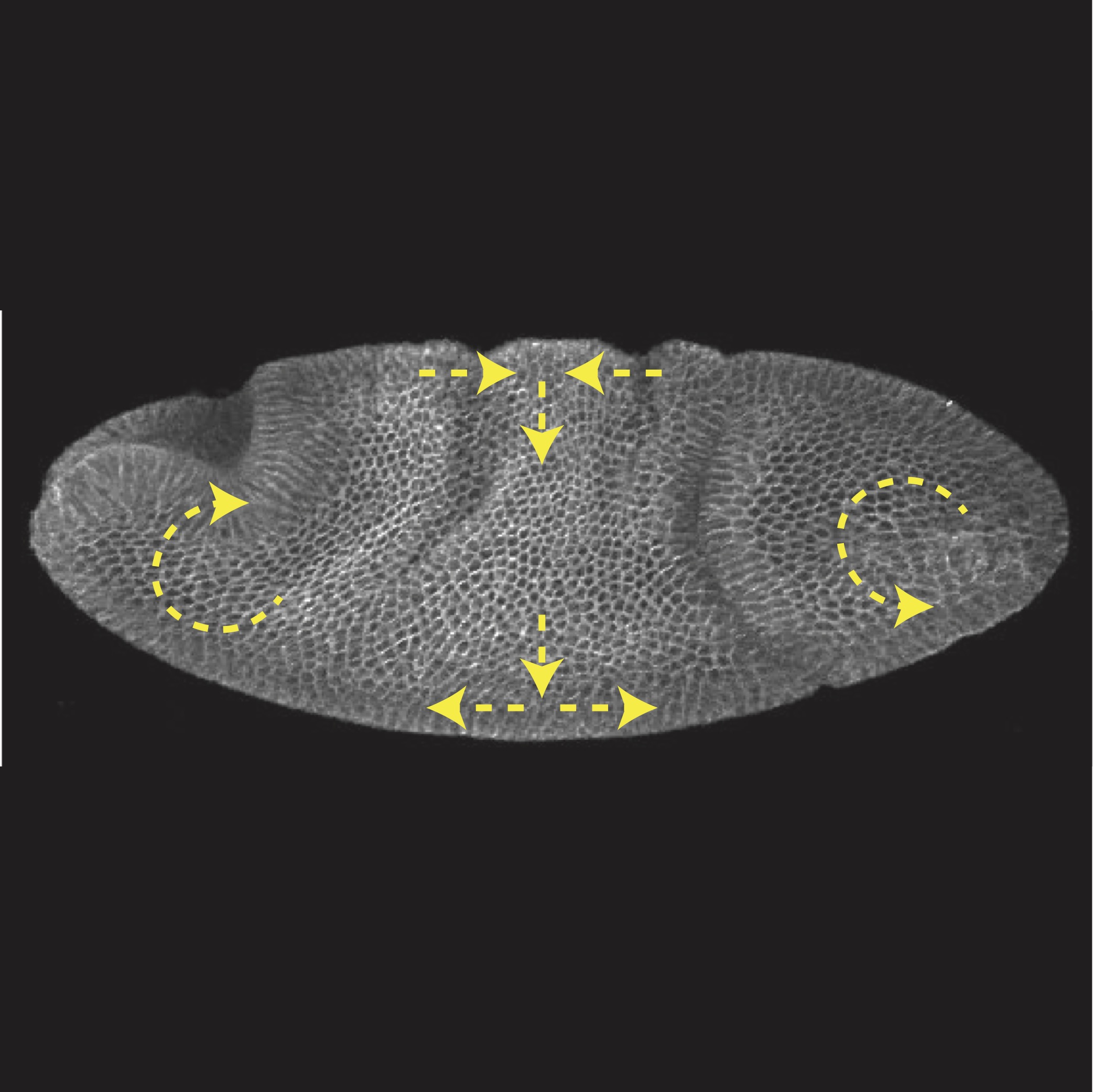
Curvature gradient drives polarized tissue flow in the Drosophila embryo
Emily W Gehrels, Bandan Chakrabortty, Marc-Eric Perrin, Matthias Merkel, Thomas Lecuit
Proceedings of the National Academy of Sciences of the United States of America 120 (2023)10.1073/pnas.2214205120
Enhanced cell viscosity: a new phenotype associated with lamin A/C alterations
Cécile Jebane, Alice-Anaïs Varlet, Marc Karnat, Lucero Hernandez- Cedillo, Amélie Lecchi, Frédéric Bedu, Camille Desgrouas, Corinne Vigouroux, Marie-Christine Vantyghem, Annie Viallat, Jean-François Rupprecht, Emmanuèle Helfer, Catherine Badens
iScience 26:107714 (2023)10.1016/j.isci.2023.107714
Physical mechanisms of red blood cell splenic filtration
Alexis Moreau, François Yaya, Huije Lu, Anagha Surendranath, Anne Charrier, Benoit Dehapiot, Emmanuèle Helfer, Annie Viallat, Zhangli Peng
Proceedings of the National Academy of Sciences of the United States of America (2023)10.1101/2023.01.10.523245
Analytical theory for a droplet squeezing through a circular pore in creeping flows under constant pressures
Zhengxin Tang, François Yaya, Ethan Sun, Lubna Shah, Jie Xu, Annie Viallat, Emmanuèle Helfer, Zhangli Peng
Physics of Fluids 35:082016 (2023)10.1063/5.0156349
2022
Dynamics of Individual Red Blood Cells Under Shear Flow: A Way to Discriminate Deformability Alterations
Scott Atwell, Catherine Badens, Anne Charrier, Emmanuèle Helfer, Annie Viallat
Frontiers in Physiology 12 (2022)10.3389/fphys.2021.775584
Mechanochemical Principles of Spatial and Temporal Patterns in Cells and Tissues
Anaïs Bailles, Emily Gehrels, Thomas Lecuit
Annual Review of Cell and Developmental Biology 38 (2022)10.1146/annurev-cellbio-120420-095337
Programming Directed Motion with DNA-Grafted Particles
Emily W Gehrels, W. Benjamin Rogers, Zorana Zeravcic, Vinothan N Manoharan
ACS Nano 16:9195-9202 (2022)10.1021/acsnano.2c01454
Longitudinal to Transverse Metachronal Wave Transitions in an In Vitro Model of Ciliated Bronchial Epithelium
Olivier Mesdjian, Chenglei Wang, Simon Gsell, Umberto D’ortona, Julien Favier, Annie Viallat, Etienne Loiseau
Physical Review Letters 129:038101 (2022)10.1103/PhysRevLett.129.038101
Lrrcc1 and Ccdc61 are conserved effectors of multiciliated cell function
Aude Nommick, Camille Boutin, Olivier Rosnet, Claire Schirmer, Elsa Bazellières, Virginie Thomé, Etienne Loiseau, Annie Viallat, Laurent Kodjabachian
Journal of Cell Science (2022)10.1242/jcs.258960
Industrial Production of Red Blood Cells from Stem Cells: A Vision Finally within Reach
Guillaume Rousseau, Fanny Rasschaert, Mathilde Maestrali, George Lardier, Florent Mathieu, Marie-Catherine Giarratana, Pierre Loche, François Yaya, Christelle Mazurier, Philippe Le Mignot, Laetitia Houx, Coralie Sabin, Annie Viallat, Luc Douay
Blood 140:4691-4692 (2022)10.1182/blood-2022-158186
2020
Mechanical adaptation of monocytes in model lung capillary networks
Jules Dupire, Pierre-Henri P Puech, Emmanuèle Helfer, Annie Viallat
Proceedings of the National Academy of Sciences of the United States of America 117:14798 (2020)
Hydrodynamic model of directional ciliary-beat organization in human airways
Simon Gsell, Etienne Loiseau, Umberto D’ortona, Annie Viallat, Julien Favier
Scientific Reports 10 (2020)10.1038/s41598-020-64695-w
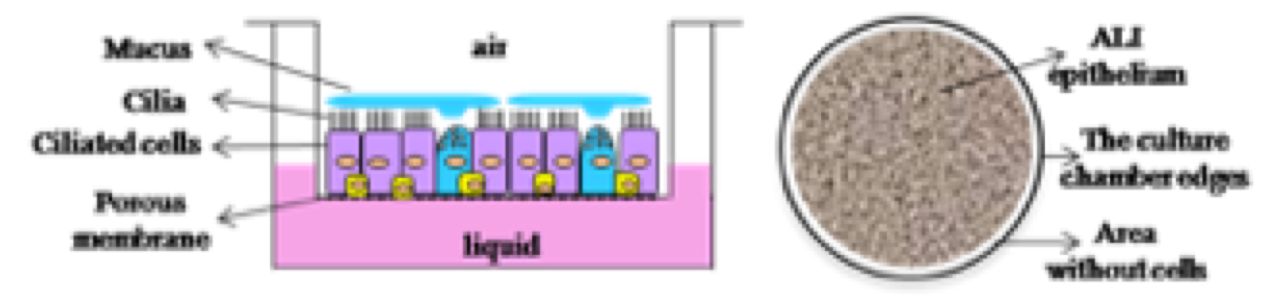
Active mucus–cilia hydrodynamic coupling drives self-organization of human bronchial epithelium
Etienne Loiseau, Simon Gsell, Aude Nommick, Charline Jomard, Delphine Gras, Pascal Chanez, Umberto D’ortona, Laurent Kodjabachian, Julien Favier, Annie Viallat
Nature Physics (2020)10.1038/s41567-020-0980-z
2019
Self-organization of red blood cell suspensions under confined 2D flows
Cécile Iss, Dorian Midou, Alexis Moreau, Delphine Held, Anne Charrier, Simon Mendez, Annie Viallat, Emmanuèle Helfer
Soft Matter 2971-2980 (2019)10.1039/C8SM02571A
2018
Reconstitution of composite actin and keratin networks in vesicles
J. Deek, R. Maan, E. Loiseau, R. Bausch
Soft Matter 14:1897-1902 (2018)10.1039/c7sm00819h
Using DNA strand displacement to control interactions in DNA-grafted colloids
Emily W. Gehrels, W. Benjamin Rogers, Vinothan N. Manoharan
Soft Matter 14:969-984 (2018)10.1039/C7SM01722G
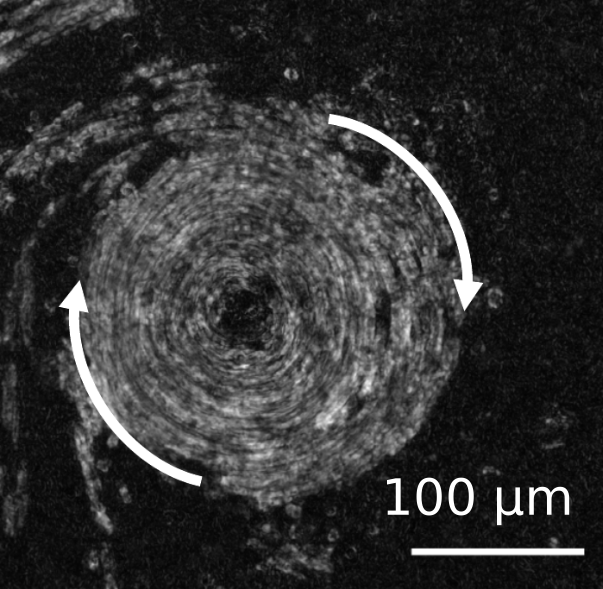
Spatiotemporal organization of cilia drives multiscale mucus swirls in model human bronchial epithelium
Mustapha-Kamel Khelloufi, Etienne Loiseau, Marc Jaeger, Nicolas Molinari, Pascal Chanez, Delphine Gras, Annie Viallat
Scientific Reports 8:2447 (2018)10.1038/s41598-018-20882-4
Adhesion of Active Cytoskeletal Vesicles
Renu Maan, Etienne Loiseau, Andreas Bausch
Biophysical Journal 115:2395-2402 (2018)10.1016/j.bpj.2018.10.013
When giant vesicles mimic red blood cell's dynamics: swinging of two-phase vesicles in shear flow
Simon Tusch, Etienne Loiseau, Al-Hair Al-Halifa, Kamel Khelloufi, Emmanuèle Helfer, Annie Viallat
Physical Review Fluids 3 (2018)10.1103/PhysRevFluids.3.123605
2017
High Aspect Ratio Sub-Micrometer Channels Using Wet Etching: Application to the Dynamics of Red Blood Cell Transiting through Biomimetic Splenic Slits
Priya Gambhire, Scott Atwell, Cécile Iss, Frédéric Bedu, Igor Ozerov, Catherine Badens, Emmanuèle Helfer, Annie Viallat, Anne Charrier
Small 13:1700967 (2017)10.1002/smll.201700967
2016
Shape remodeling and blebbing of active cytoskeletal vesicles
Etienne Loiseau, Jochen A. M. Schneider, Felix C. Keber, Carina Pelzl, Gladys Massiera, Guillaume Salbreux, Andreas R. Bausch
Science Advances 2:UNSP e1500465 (2016)10.1126/sciadv.1500465
• ANR, 2023-27 Natural and assisted airways drainage in COPD